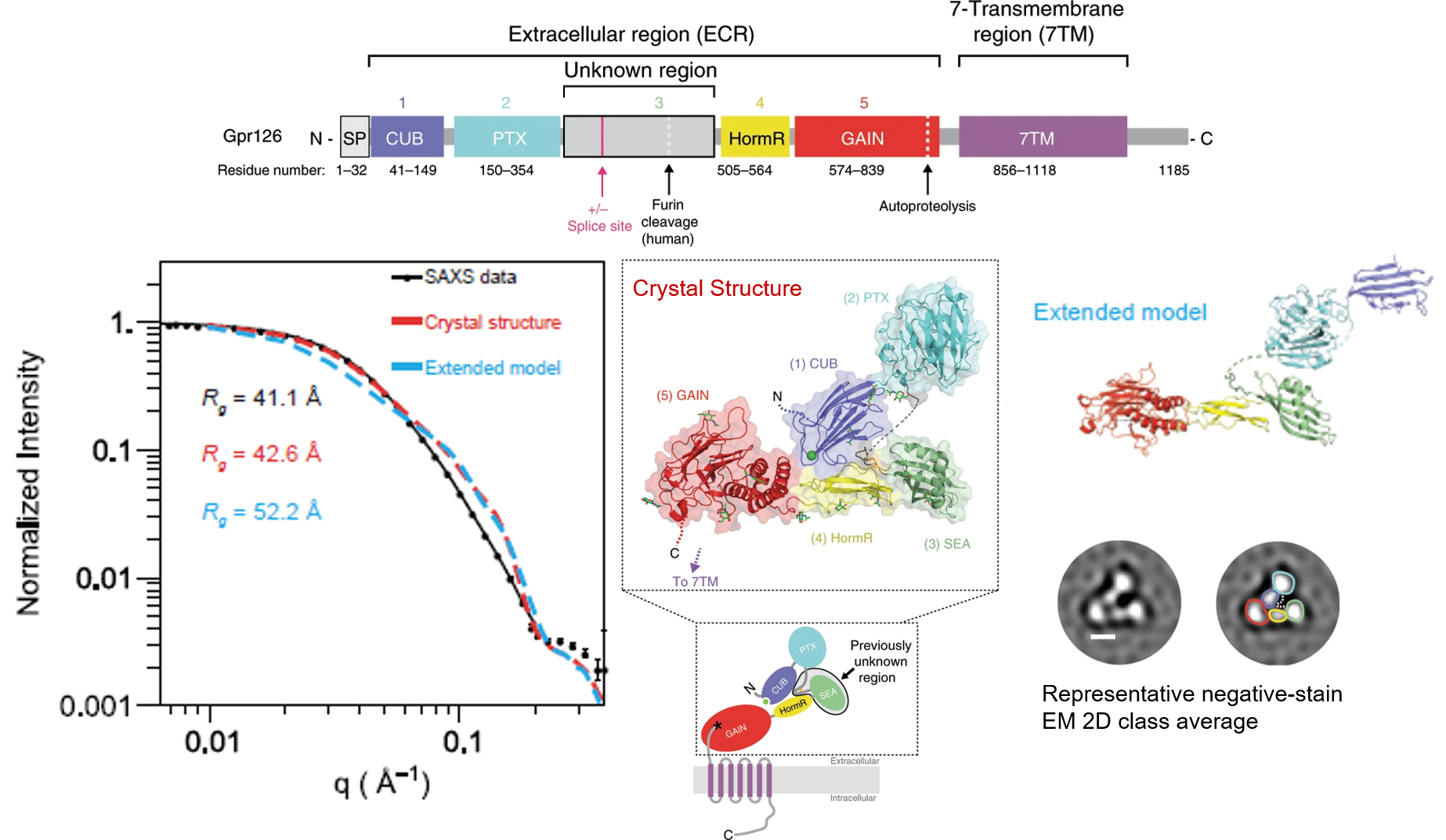
Cellular communication mediated by a variety of cell-surface receptors involves ligand induced conformational changes in the extracellular region (ECR). A variety of drugs such as cetuximab (Epidermal Growth Factor Receptor), etrolizumab (Integrins), and erenumab (calcitonin receptor-like receptor) function by trapping ECRs in specific conformations and have proved to be effective therapeutic agents in several cancers, bowel diseases, and migraine. Leon et al., have addressed a class of relatively understudied G-protein couple receptors (GPCRs) called adhesion-GPCRs (aGPCRs) which have a structurally unique ECR with a diverse set of mechanistic possibilities. Specifically, they study the Gpr126, which is known to be essential for Schwann cell myelination, involved in heart development in the mouse model, and inner ear development in Zebra fish. Determination of the high-resolution structure of the Zebra fish Gpr126 ECR revealed the involvement of a heretofore undefined domain that has splice variants with or without a 23aa stretch in different isotypes (-ss or +ss) and is the primary determinant of whether the ECR is in the open active state or the closed inactive state. Also remarkable was the calcium dependent site at the tip of the ECR which promoted the closed inactive state of Gpr126. The closed conformation observed in the crystallization studies was confirmed using a combination of negative staining EM and SAXS. SEC-SAXS performed at BioCAT showed that the radius of gyration (Rg) of the crystallization construct was ~42 Å as opposed to ~ 52 Å characteristic of the open conformation. Negative stain EM 2D class averages for the +ss isoform showed a much higher degree of conformational diversity compared to the -ss construct, a significant fraction of which were in open conformations strongly suggestive of the role of the 23aa insertion in destabilizing the closed conformation. Rg values obtained using SAXS with the +ss constructs were predictably larger than that for the -ss construct and elution volumes in size-exclusion chromatography also strongly suggest a more extended conformation for the +ss construct. Furthermore, measuring cAMP levels which is a byproduct of a well-known G protein signaling assay showed that the +ss construct does indeed have measurably higher activity than the -ss isotype. Leon et al have used a multi-technique approach to showcase the structure based mechanistic complexity involved in the aGPCR ECRs, which presents them as drug targets with immense potential.
See: Leon K, Cunningham RL, Riback JA, Feldman E, Li J, Sosnick TR, Zhao M, Monk KR, Araç D. “Structural basis for adhesion G protein-coupled receptor Gpr126 function.” Nat Commun. 2020 Jan 10;11(1):194. doi: 10.1038/s41467-019-14040-1. PMID:31924782